 Monitor patients frequently for signs and symptoms of neurological impairment. Optimal timing between the administration of XARELTO ® and neuraxial procedures is not known.A history of spinal deformity or spinal surgery.A history of traumatic or repeated epidural or spinal punctures.Concomitant use of other drugs that affect hemostasis, such as non-steroidal anti-inflammatory drugs (NSAIDs), platelet inhibitors, other anticoagulants, see Drug Interactions.Factors that can increase the risk of developing epidural or spinal hematomas in these patients include: 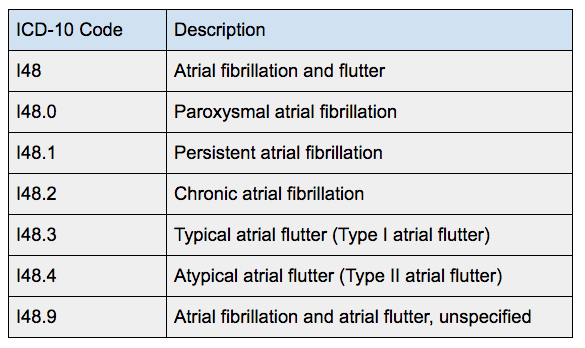
Consider these risks when scheduling patients for spinal procedures. These hematomas may result in long-term or permanent paralysis. If anticoagulation with XARELTO ® is discontinued for a reason other than pathological bleeding or completion of a course of therapy, consider coverage with another anticoagulant.Įpidural or spinal hematomas have occurred in patients treated with XARELTO ® who are receiving neuraxial anesthesia or undergoing spinal puncture. Premature discontinuation of any oral anticoagulant, including XARELTO ®, increases the risk of thrombotic events. 
Premature discontinuation of XARELTO ® increases the risk of thrombotic events WARNING: (A) PREMATURE DISCONTINUATION OF XARELTO ® INCREASES THE RISK OF THROMBOTIC EVENTS,Ī. 
XARELTO ® is indicated for thromboprophylaxis in pediatric patients aged 2 years and older with congenital heart disease who have undergone the Fontan procedure. XARELTO ® is indicated for the treatment of venous thromboembolism (VTE) and reduction in the risk of recurrent VTE in pediatric patients from birth to less than 18 years after at least 5 days of initial parenteral anticoagulant treatment. XARELTO ®, in combination with aspirin, is indicated to reduce the risk of major thrombotic vascular events (myocardial infarction, ischemic stroke, acute limb ischemia, and major amputation of a vascular etiology) in adult patients with peripheral artery disease (PAD), including patients who have recently undergone a lower extremity revascularization procedure due to symptomatic PAD. XARELTO ®, in combination with aspirin, is indicated to reduce the risk of major cardiovascular events (cardiovascular death, myocardial infarction, and stroke) in adult patients with coronary artery disease (CAD). XARELTO ® is indicated for the prophylaxis of venous thromboembolism (VTE) and VTE-related death during hospitalization and post hospital discharge in adult patients admitted for an acute medical illness who are at risk for thromboembolic complications due to moderate or severe restricted mobility and other risk factors for VTE, and not at high risk of bleeding. XARELTO ® is indicated for the prophylaxis of DVT, which may lead to PE in adult patients undergoing knee or hip replacement surgery. XARELTO ® is indicated for the reduction in the risk of recurrence of DVT and/or PE in adult patients at continued risk for recurrent DVT and/or PE after completion of initial treatment lasting at least 6 months. XARELTO ® is indicated for the treatment of pulmonary embolism (PE). 
XARELTO ® is indicated for the treatment of deep vein thrombosis (DVT). There are limited data on the relative effectiveness of XARELTO ® and warfarin in reducing the risk of stroke and systemic embolism when warfarin therapy is well controlled. XARELTO ® (rivaroxaban) is indicated to reduce the risk of stroke and systemic embolism in adult patients with nonvalvular atrial fibrillation (AF). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
